
Gene set enrichment analysis (GSEA) was performed to analyze 9 glycolysis-related gene sets, which are implicated in the glycolysis process. A total of 12 common types of solid tumors were included in the current study. 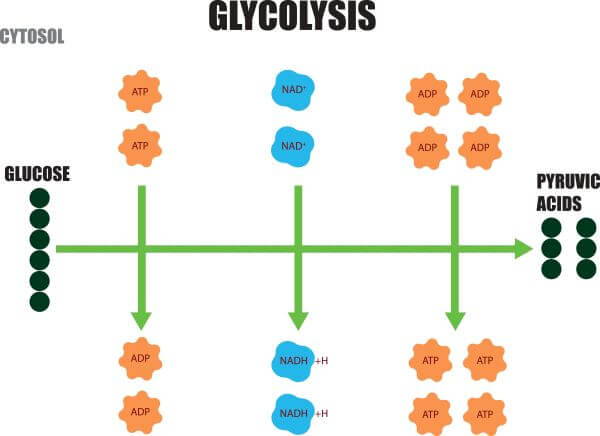
The current study reconstructed the metabolic pattern in progression of cancer to identify genetic changes specific in cancer cells. These changes promotes rapid cell proliferation and tumor growth, which is further referenced a process known as the Warburg effect. Metabolism of tumor cells is characterized by abnormal increase in anaerobic glycolysis, regardless of high oxygen concentration, resulting in a significant accumulation of energy from glucose sources. Would the addition of mitochondria to ruptured red blood cells switch their glycolytic pathway from lactate- to pyruvate-producing one? The time has arrived to reexamine the original, dogmatic separation of glycolysis into two distinct pathways and put to test the hypothesis of a unified, singular pathway, the end-product of which is lactate, the real substrate of the mitochondrial TCA cycle.Metabolic pattern reconstruction is an important factor in tumor progression. For example, pyruvate has been the substrate of choice in measuring with an oxygen electrode the rate of mitochondrial State III respiration, while lactate has never been tried as a possible alternative substrate. Amazingly, several possible basic experiments have yet to be performed that could eliminate the doubts still surrounding lactate and its mitochondrial oxidative utilization. Nonetheless, would MAS really be necessary for that function if glycolysis always proceeds to the end-products, lactate and NAD+? An additional dilemma the originally proposed aerobic glycolysis presents has to do with the glycolytic pathway of erythrocytes, which despite its highly aerobic environment, always produces lactate as its end-product. The malate-aspartate shuttle (MAS), a major redox shuttle in the brain, was proposed as an alternative pathway for NAD+ generation for aerobic glycolysis. These findings have shed light on a major drawback of the originally proposed aerobic version of the glycolytic pathway i.e., its inability to regenerate NAD+, as opposed to anaerobic glycolysis that features the cyclical ability to regenerate NAD+ upon pyruvate reduction to lactate by the cytosolic form of LDH. The mitochondrial form of the enzyme oxidizes lactate to pyruvate and concomitantly produces the reducing agent NADH. Direct and indirect evidence now indicate that the enzyme lactate dehydrogenase (LDH) resides not only in the cytosol, as part of the glycolytic pathway machinery, but also in the mitochondrial outer membrane. In the past two decades monocarboxylate transporters (MCTs) were identified and localized in both cellular and mitochondrial membranes. These findings were met with great skepticism, doubting the idea that lactate could play any role in bioenergetics, although multiple studies from the 1920s and 1930s reveal that researchers at the time were aware of the ability of brain tissue to oxidize it, a process they assumed to be a lactate-clearing mechanism. 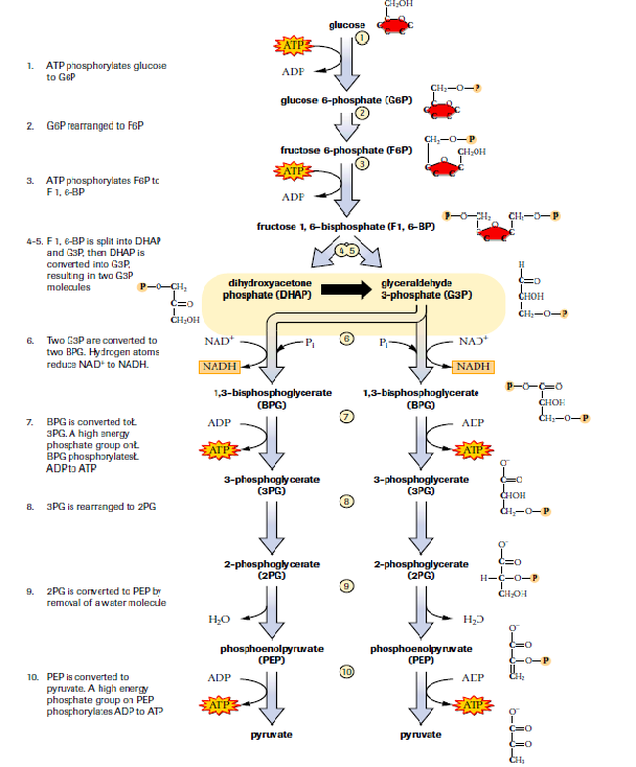
Several studies in the 1980s have shown that both muscle and brain tissue are capable of oxidizing and utilizing lactate as an energy substrate, challenging this monocarboxylate’s reputation as a useless end-product of anaerobic glycolysis. Accordingly, two outcomes of this pathway were proposed, an aerobic glycolysis, with pyruvate as its final product, and an anaerobic glycolysis, identical to the aerobic one, except for an additional step, where pyruvate is reduced to lactate. For more than seven decades, it has been taught precisely the way its sequence was proposed by Embden, Meyerhof and Parnas. Glycolysis, the pathway of enzymatic reactions responsible for the breakdown of glucose into two trioses and further into pyruvate or lactate, was elucidated in 1940.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
